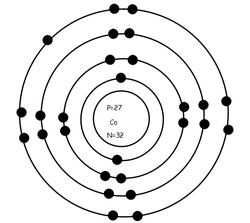 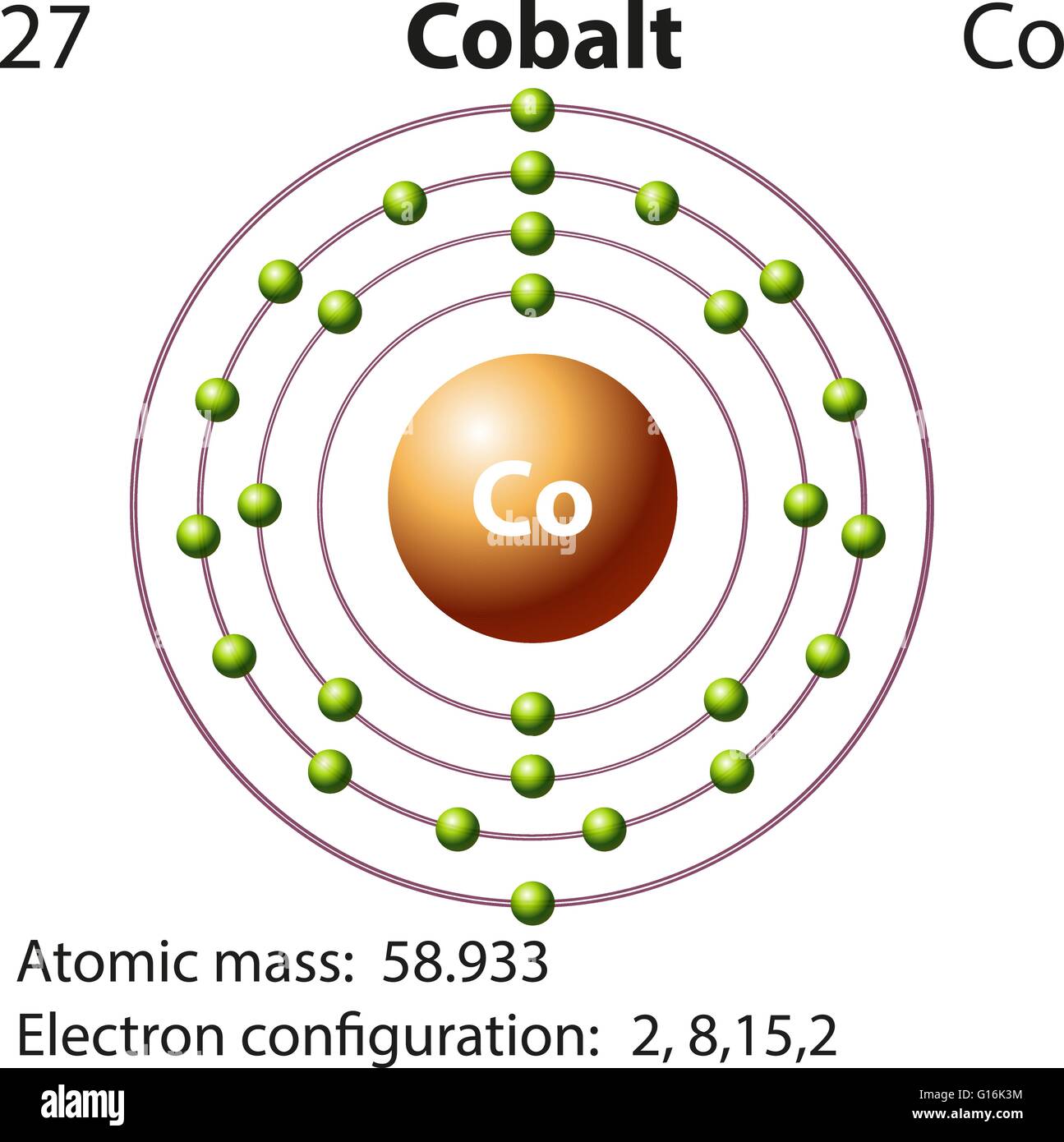
The remaining #"7 e"^(-)# will now be distributed in the 3d-subshell. Co (Cobalt) is an element with position number 27 in the periodic table. Because the 4s orbital is filled before the 3d-orbitals, the next two electrons are going to be distributed on the fourth energy level Taking it one subshell at a time, you will have #color(red)(cancel(color(black)(n=3 -> "17 e"^(-))))# in the #3s#, #3p#, and #3d# subshellsĪnd conclude that the electrons that surround the nucleus of a cobalt atom are spread out on #3# energy levels. Chemical Properties of Cobalt Electrochemical Equivalent: 1.0994g/amp-hr Electron Work Function: 5eV Electronegativity: 1.88 (Pauling) 1.7 (Allrod Rochow). The third energy level can hold #"18 e"^(-)#, so in theory it can hold the remaining This means that you must fill the 4s-orbital first, then distribute the rest of the electrons to the 3d-orbitals. Now, it's very important to remember that when you're adding electrons to an atom, the 3d-orbitals, which are located on the third energy level, are higher in energy than the 4s-orbital. These electrons will be placed in orbitals in order of increasing energy in accordance to the Aufbau Principle. The two outermost electrons are the 4s electrons so these are the two electrons that most likely will be lost. In your case, cobalt, #"Co"#, is said to have a total of #27# electrons surrounding its nucleus. Co(2+) would most likely be 1s2 2s2 2p6 3s2 3p6 4s0 3d7, and Co would have 4s2 instead (the rest otherwise the same). You can use this equation to find the maximum number of electrons that can be added to each energy level. Cobalt is a chemical element with the symbol Co and atomic number 27. #color(blue)(|bar(ul(color(white)(a/a)"no. Electron configuration 3d 7 4s 2: Electrons per shell: 2, 8, 15, 2: Physical properties. The relationship that exists between the energy level, #n#, and the number of electrons it can hold can be written like this Phenom., 1980, 21, 275.The number of electrons each energy level can hold increases as you add more and more energy levels to an atom. But, the n + l n + l rule, as many other rules of old quantum theory, is not 100 working, and thus, sometimes gives wrong electron configuration. Mårtensson, "Core-Level Binding Energies in Metals," J. Thus, the electron configuration of Mn M n is Ar3d54s2 A r 3 d 5 4 s 2 while that of Co C o is Ar3d74s2 A r 3 d 7 4 s 2. formed is Fe(H 2O) 5 NO2+ Pentamminenitrito-N-cobalt (III) chloride. Electron configuration can be done in two. So, configuration of outermost electrons are 28Ni 3d 4s (From CO) ×× 3d 4p 24. Lide, (Ed.) in Chemical Rubber Company handbook of chemistry and physics, CRC Press, Boca Raton, Florida, USA, 81st edition, 2000. The electron configuration of cobalt is 1s2 2s2 2p6 3s2 3p6 3d7 4s2, if the electron arrangement is through orbitals. In chemistry, the noble gas configuration is a shorthand method of writing an atom’s electron configuration.The reason for using the noble gas configuration is because the full electron configuration becomes very long for atoms with high atomic numbers. Ley, Eds., Photoemission in Solids I: General Principles (Springer-Verlag, Berlin) with additional corrections, 1978. The noble gas configuration is a shorthand electron configuration for atoms. Burr, "Reevaluation of X-Ray Atomic Energy Levels," Rev. They are tabulated elsewhere on the WWW (reference 4) and in paper form (reference 5). Keeping in mind the quantum numbers the electron configuration of cobalt is 1s22s22p63s23p64s23d7. The data are adapted from references 1-3. I am grateful to Gwyn Williams (Jefferson Laboratory, Virginia, USA) who provided the electron binding energy data. The binding energies are quoted relative to the vacuum level for rare gases and H 2, N 2, O 2, F 2, and Cl 2 molecules relative to the Fermi level for metals and relative to the top of the valence band for semiconductors. All values of electron binding energies are given in eV. Alternatively, you can count the superscripts of the energy levels until you get to ten. Therefore, the valence electrons of cobalt are nine. As you can see, sodium has a 3s 1 that neon does not have, therefore, the noble gas configuration for sodium would be Ne3s 1. This electron configuration shows that the last shell of cobalt has two electrons and the d-orbital has a total of seven electrons. 1967, 47, 1300.Įlectron binding energies Electron binding energies for cobalt. The full electron configuration for sodium is 1s 2s 2 2 2p 6 3s 1 and neon is 1s 2s 2 2 2p 6. These effective nuclear charges, Z eff, are adapted from the following references:
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
